LFP in Different Particle Size Distributions
IBUvolt® is offered in two particle size distributions – as LFP200 and LFP400. LFP400 is a coarser form of LFP powder (d99 ~40µm) with good volumetric energy density (Wh/L), very good specific power (W/g) and excellent cycle life.
LFP200 is a finer form (d99 ~20µm) of LFP. By blending with coarser battery powders such as our LFP400, higher densification can be achieved to improve the volumetric energy density of the electrode, which can lead to battery performance benefits. The two battery materials can be used in a ratio of approximately 1:4.
| IBUvolt® LFP400 | IBUvolt® LFP200 | LFP400 : LFP200 (4:1) | |
| Particle size d50 [µm] | 11.1 | 5.8 | 8 |
| Particle size d99 [µm] | 37.9 | 19.1 | 33 |
| Tamped density [g/cm³] | 1.0 – 1.4 | 0.9 – 1.3 | 1 |
| Electrode density [g/cm³] | 2.3* | 2.1* | ≥ 2.4* |
| Specific capacity [mAh/g] | ≥ 149 | tbd | ≥ 147 |
| Energy density [Wh/l] | 1131 | tbd | > 1165 |
| Data sheet | Download | Download |
* Example for laboratory test cells
Production of Our Lithium Iron Phosphate
For more than 10 years IBU-tec has been involved in the development and production of LFP cathode material. The starting materials first pass through spray drying, are refined in a rotary kiln and then milled: an excellent production performance with high quality.
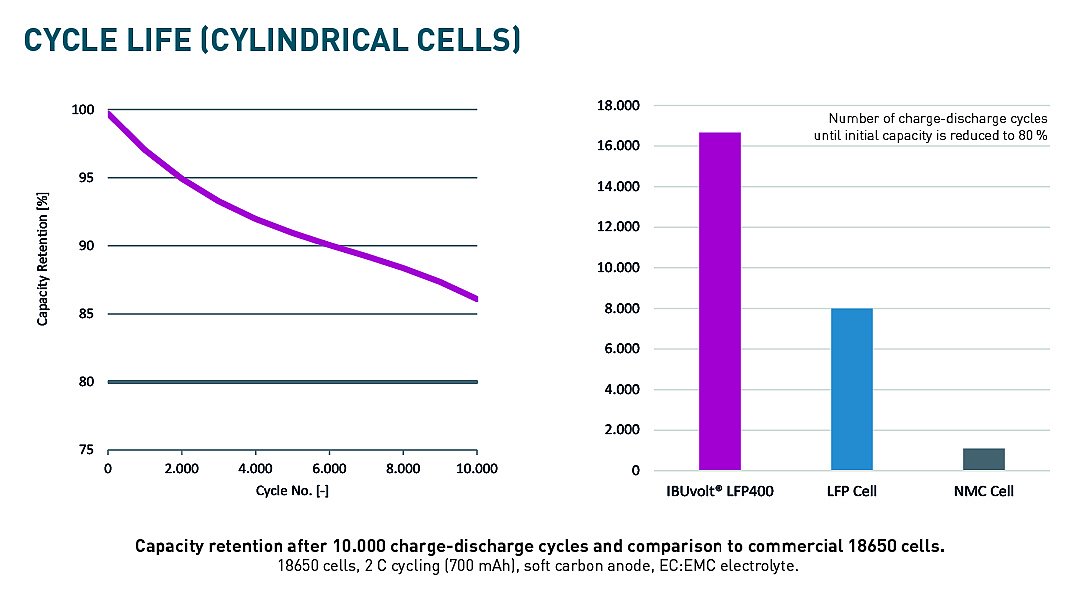
Long Life and Cycle Stability of IBUvolt® LFP
IBUvolt® LFP maintains its capacity over thousands of charging cycles – a comparatively long service life. Thus, at comparable acquisition costs, the specific costs per charging cycle or kilometer are reduced by up to 50% due to a significantly longer operating time. In addition, due to its high thermal stability, the LFP battery offers greater safety against fires than alternative cathode materials – the risk of uncontrollable battery fires is significantly reduced – a clear advantage for use in transport vehicles and large stationary energy storage systems.
Particle Size and Efficiency in Battery Production
IBUvolt® LFP has been optimized for good processability in the production of battery cells on an industrial scale. The manufacturing process produces round granules with an average particle size of about 11 µm and 6 µm for LFP400 and LFP200, respectively.
The comparatively high carbon content of 3.5 – 3.9 wt.% leads to the advantage of high electrical conductivity. This reduces the internal resistance in the battery cell and less electrical energy is converted into heat. This increases energy efficiency and safety compared to other LFP products with lower conductivity. Furthermore, the high carbon content of IBUvolt® LFP allows less conductive carbon black to be used in cathode production, which means savings in cell production costs.
Battery Charging Speed
Compared to other lithium iron phosphate brands, IBUvolt® LFP shows significantly higher rate stability during charging. Thus, higher charging currents can be used, which is especially desired for automotive applications. With prismatic test cells, more than 80% of the cell capacity could be charged within 12 min.
Background on Lithium Iron Phosphate Batteries
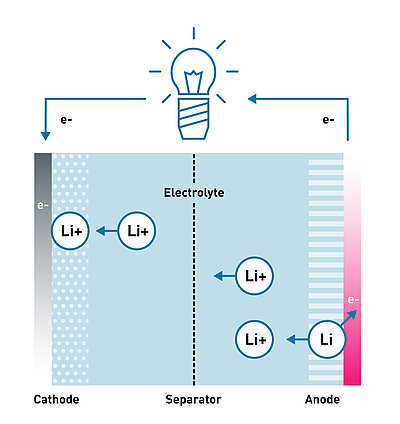
Energy storage systems are a valuable key technology for not having to rely on the use of fossil raw materials for energy production and being able to use other sources, such as wind power and photovoltaic systems. Currently available storage systems are largely based on the proven lithium-ion battery technology. Lithium-ion batteries consist of four basic components that make up the battery's cells: Cathode, Anode, Separator and Electrolyte. IBU-tec has many years of experience in the production of lithium iron phosphate cathode material (LFP or LiFePO4).
When charging a lithium-ion battery or lithium-ion accumulators, lithium ions are transported through the electrolyte layer from the cathode to the anode. In the anode, which is often made of graphite or other carbon-rich materials, the reversible intercalation of the lithium takes place with the absorption of electrons. When the battery is discharged, this process is reversed. The lithium-rich anode layer releases electrons and the resulting lithium ions are transported to the cathode. The resulting current flow can be used by electrical consumers. This cycle can be repeated several times in rechargeable batteries, although certain memory effects are inevitable. The higher the quality of the battery and the materials used in it, such as lithium iron phosphate, the longer the service life can be.
Lithium iron phosphate is becoming increasingly important as a cathode material in modern batteries. Depending on the end application and field of use – be it in the field of e-mobility or in stationary energy storage – different requirements arise.
The designation LFP is derived from the empirical formula LiFePO4. LFP cathodes are free of the heavy metal nickel and the critical raw material cobalt. Compared to other cathode materials, LFP also shows advantages in durability and safety.