| Zinc(II)-Oxide 60 | Zinc(II)-Oxide 35 | Zinc(II)-Oxide 20 | |
| Specific surface area | 60 ± 5 m2/g | 35 ± 2 m2/g | 20 ± 2 m2/g |
| Primary particle size | 10 – 15 nm | 20 – 25 nm | 20 – 50 nm |
| Agglomerate size | 0.2 – 0.3 µm | 0.3 – 0.4 µm | 0.4 – 0.5 µm |
| Purity | > 99 wt.% | > 99 wt.% | > 99 wt.% |
| Bulk density | 80 kg/m3 | 90 – 100 kg/m3 | 100 kg/m3 |
| Crystallographic phase | Zincite | Zincite | Zincite |
| Possible applications |
|
|
|
| Datasheet | Download | Download | Download |
Transparent Zinc Oxide in UV Protection Products
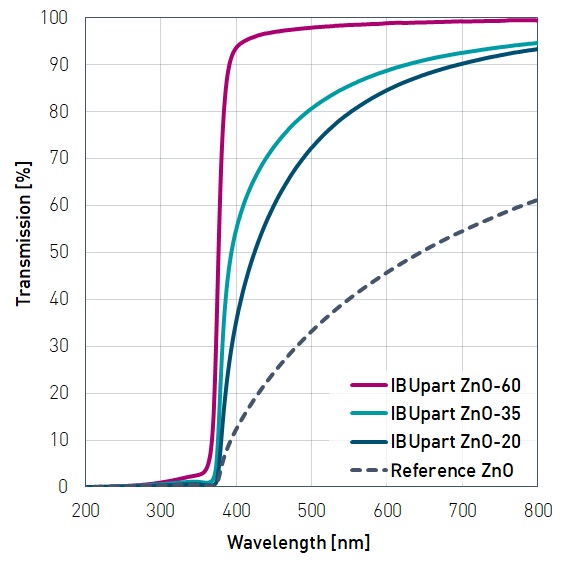
Zinc oxide shows high absorption of light over the entire UV range. This makes the material suitable for use as a sunscreen in cosmetics, paints and coatings. Dispersed ZnO nanoparticles with a size below 40 nm are transparent to visible light and can be used in clear UV protective coatings.
Traditionally used UV protection solutions reach their limits with increasing requirements. Zinc oxide can be used as a UV protection that covers the complete UV-C to UV-A range, can be used as a broadband filter and is photocatalytically inactive and thus stable over a longer period of time.
The major disadvantage of zinc oxide is the formation of an opaque layer after drying, which is why it has not enjoyed widespread acceptance as a component of UV protection products to date. We have countered this by reducing the particle size into the nanometer range. Agglomerates from the primary particles reach sizes well above 120nm, which are established as safe, but retain exceptional transparency in the visible radiation spectrum.
Full UV protection is achieved even at a low concentration, which is maintained under good dispersion properties in water and organic solvents even when converted into a suspension.
Other Uses of Zinc Oxide
Zinc oxide has special mechanical, electrical, piezoelectric and optical properties, with which it is often used as an additive in the manufacture of rubber or ceramics. Another area is its use as a pigment or as an ingredient in pharmaceutical products.
In electrical engineering, ZnO-based products are used for varistors and sensors. In addition, n-type semiconductors are created by doping zinc oxide substrates. These can be used for solar cells or LCD displays.
As an aggregate for concrete, zinc oxide reduces the hardening time and improves the hydraulic properties of the building material. In the vulcanization of tires, ZnO serves as a catalyst and to improve heat transport in the tire, which increases its service life. The glass and ceramics industries use zinc oxide as an ingredient in glazes, where it mainly serves as a flux and creates a shiny surface.
Zinc oxide also serves the chemical industry as a catalyst in a variety of reactions. Methanol synthesis from synthesis gas, for example, one of the most important basic processes in the chemical industry, uses a system of Cu-Zn-Al oxide.
Production of Zinc Oxide
At IBU-tec, nanoscale zinc oxide is produced by flame aerosol processes, for which we use our pulsation reactors. The production process delivers controllable particle sizes in a narrow distribution. Compared to nanoparticle synthesis by sol-gel processes, fewer process steps are necessary and no aqueous waste is produced.